
June Almeida And The First “Coronavirus” EM Images (1967)
In 1967, June Almeida presented a paper supposedly showing the first EM images of the “coronavirus” structure. These images were published two years after D.A. Tyrrell discovered the B814 strain in 1965 and a year after Dorothy Hamre discovered strain 229E in 1966. The samples that Almeida used to characterize the previously unseen “viruses” were provided by both Tyrrell and Hamre as neither had actually visualized the “viruses” they had already decided (assumed) were within their organ and tissue cultures. In this paper which she co-authored with Tyrrell, Almeida claimed to use a new negative staining approach for her electron microscope images on unpurified samples in order to characterize the three uncharacterized “viruses.” This is how her method is described:
Seeing SARS – the Remarkable Story of June Almeida
Development of a Novel Microscopy Technique
“Early on in her career, Almeida worked as an electron microscopy technician at the Ontario Cancer Institute in Canada. There she developed a novel imaging technique for the detection of viruses, known as immune electron microscopy (IEM) (Almeida 2008). This technique involves the detection of molecules at the ultrastructural level by labeling them with specific antibodies attached to colloidal gold particles. When a beam of electrons encounters the colloidal gold particles on an antibody bound to its antigen an electron-dense label is produced. The electron beam is then scanned and the position of the beam, combined with the intensity of the signal, is used to produce an image, referred to as an electron micrograph. This allows detection and visualization of the colloidal gold particles and therefore the target molecule. If the antigen is located on the surface of the cell, scanning electron microscopy (SEM), where the beam of electrons is streamed across the surface of a sample can be used to visualize it.”
Identification of a New Type of Virus
Tyrrell’s team of researchers had collected samples of a flu-like virus from a sick schoolboy. However, they were having trouble cultivating this virus using traditional methods, leading them to suspect that this might be a completely new type of virus (Broklehurst 2020).
Tyrrell sent some of these samples to Almeida in the hopes that she would be able to identify the virus using the IEM technique. Almeida was able to successfully produce clear images of the virus and noted that it resembled two similar viruses she had previously imaged while studying bronchitis in chickens and murine hepatitis (Brocklehurst 2020). These had previously been dismissed as poor images of influenza viruses. However, the images of the virus in the samples from the schoolboy combined with the images from Almeida’s previous work convinced Almeida and Tyrrell that these were in fact a new family of viruses.
https://www.bio-rad-antibodies.com/blog/the-remarkable-story-of-June-Almeida.html

Ignoring the rather ridiculous title as “SARS” wasn’t a “thing” until 2003, it is stated that Almeida created a new EM technique requiring gold labels and specific antibodies to interact and stain the particles she wanted to identify as “viruses.” However, without having first purified, isolated, and characterized the “virus,” it would be impossible to know which antibodies are specific to it. On top of that, antibodies are, contrary to popular belief, regularly non-specific and bind to unwanted targets. Like “viruses,” they have never been properly purified/isolated, characterized, and proven to exist. Antibodies are an entirely theoretical concept based on chemical reactions for which the antibodies are assumed to exist within. One can not use one theoretical concept (antibodies) in order to prove the existence of another theoretical concept (“viruses”).
While Almeida’s new EM technique requires leaps in logic to accept, what is even more concerning is her and Tyrrell’s attempts to convince the scientific community that the same particles identified as influenza are in fact an entirely new “virus ” Not only did they have what they considered an influenza-like “virus,” they had particles that looked exactly the same. In fact, Almeida’s own paper was rejected by peer review as the images were described as “bad pictures of influenza particles:”
The woman who discovered the first coronavirus
“However, volunteer studies demonstrated its growth in organ cultures and Dr Tyrrell wondered if it could be seen by an electron microscope.
They sent samples to June Almeida who saw the virus particles in the specimens, which she described as like influenza viruses but not exactly the same.
She identified what became known as the first human coronavirus.
Mr Winter says that Dr Almeida had actually seen particles like this before while investigating mouse hepatitis and infectious bronchitis of chickens.
However, he says her paper to a peer-reviewed journal was rejected “because the referees said the images she produced were just bad pictures of influenza virus particles”.
The new discovery from strain B814 was written up in the British Medical Journal in 1965 and the first photographs of what she had seen were published in the Journal of General Virology two years later.”
https://www.bbc.com/news/uk-scotland-52278716

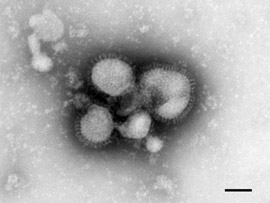
It is apparent that Almeida’s images came from a new untested method which relied on theoretical particles to define other theoretical particles. She picked those that resembled the influenza “virus” as well as those which she had seen in murine and avian diseases. You will find after reading her paper, that Almeida did so using unpurified samples which she claimed was made possible due to her own earlier research. Based on the above, it would be easy to outright reject the claims that these images are particles relating to new “viruses” without even reading her work, yet here is the full paper for those wanting all of the juicy details:
The Morphology of Three Previously Uncharacterized Human Respiratory Viruses that Grow in Organ Culture
SUMMARY
A simple method is described for examining organ cultures by electron microscopy for the presence of virus particles. The method was used to detect the presence of three hitherto uncharacterized viruses. Two of these have particles resembling those of infectious bronchitis of chickens and the third morphologically resembles the parainfluenza group of viruses.
INTRODUCTION
Organ cultures of respiratory epithelium provide a practical and sensitive means of propagating human respiratory viruses. Some viruses can at present only be grown in this way (Tyrrell & Bynoe, 1965, 1966; Hoorn & Tyrrell, 1966). Unfortunately, several such viruses can only be detected by inoculating volunteers. It was therefore decided to try to detect these viruses and characterize them morphologically by the electron-microscope technique of negative staining. Originally negative staining was applied to purified preparations of virus particles (Brenner &Horne, 1959), but it was later shown that crude preparations of whole cells could also be used to study cell-associated viruses (Horne& Nagington, 1959; Almeida & Howatson, 1963; Parsons, 1963).
In the present instance, suspensions obtained by mild treatment of the organ cultures in a glass homogenizer have been used for microscopy and three uncharacterized respiratory viruses have been seen. Two of these were of a morphological type not previously associated with human disease.
METHODS
Organ culture. Nasal epithelium and trachea were dissected from human embryos of 14 to 24 weeks gestation and planted on scratched areas of plastic Petri dishes 60 mm. diameter AA grade (Esco Rubber Co.). The nasal epithelium was supplied with 2 ml. of Eagle’s medium containing 0-2% (w/v) bovine plasma albumin; the trachea received 1.25 ml. The dishes were incubated at 33 ° in sealed humidified plastic boxes in an atmosphere of 5% (v/v) carbon dioxide in air. After 2 days’ incubation, the medium was changed and the cultures inoculated by dropping 0.2 ml. of virus suspension on to the tissue. The cultures were then incubated for a further 4 days, when the medium was removed and, where possible, titrated for virus in roller tube tissue cultures incubated at 33°.
Electron microscopy. The tissue fragments were treated very lightly in a loosely fitting glass homogenizer of the TenBroeck type with a few drops of distilled water. Most of the tissue fragment remained intact and mainly the superficial, virus-infected cells were detached. A drop of this cell suspension was mixed with an equal quantity of 3 % (w/v) phosphotungstic acid adjusted to pH 6.0 with potassium hydroxide. A small amount of this mixture was then placed on a carbon+formvar coated grid, excess fluid removed with filter paper and the specimen examined immediately in a Philips 200 electron microscope. If this procedure could not be carried out within a few hours of harvesting the tissue fragments, they were frozen at -70° until convenient.
Several known viruses were examined to establish the practicability of the method which was used then on three uncharacterized viruses that cause human upper respiratory disease. These are (a) strain 229 E of Hamre & Procknow (1966); (b) strain B814 (Tyrrell & Bynoe, 1965); (c) strain LAKEY (Tyrrell & Bynoe, 1966).
RESULTS
Virus particles or viral components were detected in almost all the cultures inoculated with known viruses, and in no instance in an uninoculated control. An additional control was provided by examining cultures that had been inoculated with herpes simplex and vaccinia viruses and then not incubated but held at 4°. No virus particles were seen in these preparations.
Each of the three uncharacterized human viruses revealed virus particles or viral components associated with the negatively stained cellular fragments. Strain 229 E contained particles (P1. 1, figs. 1, 2) resembling closely the particles of avian infectious bronchitis (Berry et al. 1964). The particles are pleomorphic in form and although varying somewhat in size have an average diameter of about 800 to 1200 A. The surface of the particles is covered with a distinct layer of projections roughly 200 A, long. These projections seem to have a narrow stalk just within the limit of resolution of the microscope and a ‘head’ roughly 100 A across.
Similar particles were found in organ cultures infected with the second uncharacterized strain, B814, which cannot at present be grown in tissue cultures (Tyrrell & Bynoe, 1965). The particles from this strain (P1. 1, figs. 3, 4) were indistinguishable both from those of 229 E (P1. 1, figs. 1, 2) and of avian infectious bronchitis.
The third unknown strain, LAKEY, had been obtained from the nasal washings from a patient with a cold (Tyrrell & Bynoe, 1966). It had been passed twice in organ cultures and the medium from these cultures produced colds in volunteers. On one occasion a very poor haemadsorption had been seen in a few outlying cells of a roller tube culture of rhesus monkey kidney inoculated 10 days previously with culture medium. Although this observation could not be repeated it was a clue suggesting that this might be some type of myxovirus. The electron-microscope preparation showed a great deal of helical material with a diameter of 180 A which was indistinguishable from the internal component of viruses such as the parainfluenza group (PI. 2, figs. 5, 6). Until adsorption to and elution from red cells has been definitely established it is not possible to say that it is a myxovirus, but the morphology found does establish that the virus belongs to the subgroup of compound viruses having a morphology like that of Newcastle disease virus (Waterson & Almeida, 1966).
DISCUSSION
The procedure that we have used for identifying viruses grown in organ culture is both simple and speedy. Any attempt at a conventional purification procedure from the tissue fragments would be difficult and inefficient since the amount of material available is so small and the proportion of infected ceils is so low. When the culture is handled in the way described, we believe that the cells that go into suspension and are used for electron microscopy are mainly those that are infected with virus. Virus particles were identified even when the titre in the supernatant was low.
Probably the most interesting finding from these experiments was that two human respiratory viruses, 229 E and B814. are morphologically identical with avian infectious bronchitis. Their biological properties, as far as they are known, are consistent with this. Both the human viruses are ether sensitive as is avian infectious bronchitis 229 E, have a similar size by filtration and multiply in the presence of an inhibitor of DNA synthesis. It will be interesting to compare the serology of the morphologically similar human and chicken viruses.
EXPLANATION OF PLATES
PLATE 1
Fig. 1 a, b. This type of particle was seen when organ cultures infected with strain 229E were examined by the present technique. The particles are pleomorphic, in the size range 800 to 1200 A, and are surrounded by a distinct 200 A long fringe. They are indistinguishable from the particles of avian infectious bronchitis, the only virus previously known to have this morphology.
Fig. 2. An additional single particle of strain 229E (for comparison with figs. 3 and 4).
Figs. 3 and 4. Human respiratory virus strain B814, which also revealed particles indistinguishable from avian infectious bronchitis. (Magnification see PI. 2).
PLATE 2
Fig. 5. Part of the cytoplasm of a disrupted cell containing a large quantity of helical component similar to that present in viruses such as parainfluenza (see fig. 6). In this case the virus was an uncharacterized human respiratory virus (LAKEY). Infected specimens of trachea and nasal epithelium both contained abundant viral material.
Fig. 6. Length of internal helical component from parainfluenza virus type 3 grown in organ culture of nasal epithelium for comparison with fig. 5. The bar represents 1000 A.


doi 10.1099/0022-1317-1-2-175.
In Summary:
- Almeida developed a novel imaging technique for the detection of “viruses,” known as immune electron microscopy
- This technique involved the detection of molecules at the ultrastructural level by labeling them with specific antibodies attached to colloidal gold particles (how would the “specific” antibodies be known for an non-isolated, uncharacterized “virus” from unpurified samples?)
- When a beam of electrons encounters the colloidal gold particles on an antibody bound to its antigen an electron-dense label is produced
- The electron beam is then scanned and the position of the beam, combined with the intensity of the signal, is used to produce an image, referred to as an electron micrograph
- This allows detection and visualization of the colloidal gold particles and therefore the target molecule
- Tyrrell’s team of researchers had collected samples of a flu-like “virus” from a sick schoolboy
- However, they were having trouble cultivating this “virus” using traditional methods, leading them to suspect that this might be a completely new type of “virus”
- Almeida was able to produce “clear” images of the “virus” and noted that it resembled two similar “viruses” she had previously imaged while studying bronchitis in chickens and murine hepatitis
- These had previously been dismissed and her paper, which had been sent to a peer-reviewed journal, was rejected “because the referees said the images she produced were just bad pictures of influenza “virus” particles”.
- However, the images of the “virus” in the samples from the schoolboy combined with the images from Almeida’s previous work convinced Almeida and Tyrrell that these were in fact a new family of “viruses”

- A simple method waa described for examining organ cultures by electron microscopy for the presence of “virus” particles (how would she know which ones were “virus” particles and which ones were not?)
- Two of the cultures had particles resembling those of infectious bronchitis of chickens and the third morphologically resembled the parainfluenza group of “viruses”
- Almeida claimed several “viruses” could only be detected by inoculating volunteers (what exactly were they injecting if they did not know if a “virus was present or not?)
- It was decided to try to detect these “viruses” and characterize them morphologically by the electron-microscope technique of negative staining
- Originally, negative staining was applied to purified preparations of “virus” particles, but Almeida stated it was later shown that crude preparations of whole cells could also be used to study cell-associated “viruses” based on results from her own work
Quick Detour on Almeida’s Crude Preparations Paper (1963):
A NEGATIVE STAINING METHOD FOR CELL-ASSOCIATED VIRUS
“In summary, the technique described here offers three main advantages in the examination of virus-infected cells by negative staining.
First, it is a sensitive and rapid method of establishing the presence or absence of virus in any preparation.
Second, since the material receives the minimal amount of handling, viruses retain structural details that may be lost in lengthy purification procedures.
Third, a virus can be examined in relation to other particles around it and to the constituents of the cell in which it is contained.”
doi: 10.1083/jcb.16.3.616.
According to Almeida, purification processes may destroy structural details so she wanted to have minimal handling in order to examine these unknown, uncharacterized particles in relation to the other particles around them. This raises the question, if she had never seen these “viruses” nor characterized them before, how would she know which ones are the “virus” particles she is looking for versus those that are around them? Obviously, this admittance to examining unpurified material to find the particles claimed to be “coronaviruses” immediately discredits her images.
End Detour.
- Organ culture treatment:
- Nasal epithelium and trachea were dissected from human embryos of 14 to 24 weeks gestation and planted on scratched areas of plastic Petri dishes 60 mm. diameter AA grade (Esco Rubber Co.)
- The nasal epithelium was supplied with 2 ml. of Eagle’s medium containing 0-2% (w/v) bovine plasma albumin; the trachea received 1.25 ml
- The dishes were incubated at 33° in sealed humidified plastic boxes in an atmosphere of 5% (v/v) carbon dioxide in air
- After 2 days’ incubation, the medium was changed and the cultures inoculated by dropping 0.2 ml. of “virus” suspension on to the tissue
- The cultures were then incubated for a further 4 days, when the medium was removed and, where possible, titrated for “virus” in roller tube tissue cultures incubated at 33°
- For EM imaging, a drop of cell suspension was mixed with an equal quantity of 3 % (w/v) phosphotungstic acid adjusted to pH 6.0 with potassium hydroxide
- Several known “viruses” were examined to establish the practicability of the method which was used then on three uncharacterized “viruses” that cause human upper respiratory disease
- In other words, she used this method to find particles already assumed to be “viruses”in order to state it was effective to find unknown “viruses“
- “Virus” particles or “viral” components were detected in almost all the cultures inoculated with known “viruses”
- An additional “control” was provided by examining cultures that had been inoculated with herpes simplex and vaccinia “viruses” and then not incubated but held at 4° yet no “virus” particles were seen in these preparations
- Each of the three uncharacterized human “viruses” revealed “virus” particles or “viral” components associated with the negatively stained cellular fragments
- It should be clear that a “virus” was assumed to be within the unpurified samples as no particles had been isolated nor characterized
- Hamre’s strain 229 E contained particles resembling closely the particles of avian infectious bronchitis
- Tyrrell’s strain B814 showed similar particles in organ cultures
- Tyrrell’s other unknown strain, LAKEY, had been obtained from the nasal washings from a patient with a cold and it had been passed twice in organ cultures and it was indistinguishable from the internal component of “viruses” such as the parainfluenza group
- Almeida claimed any attempt at a conventional purification procedure from the tissue fragments would be difficult and inefficient since the amount of material available is so small and the proportion of infected ceils is so low
- When the culture was handled in the way she described, she believed that the cells that went into suspension and were used for electron microscopy were mainly those that were infected with “virus”
- Almeida concluded that the most interesting finding from these experiments was that two human respiratory “viruses,” 229 E and B814, were morphologically identical with avian infectious bronchitis and that their biological properties, as far as they were known, were consistent with this


At the very least, it was extremely courteous of Almeida to admit that she did not purify nor isolate the particles assumed to be “viruses” before taking their photographs so that we can immediately dismiss the claims that these particles are in fact “viruses.” Obviously, the purification and isolation steps should have been performed first by Tyrrell and Hamre but unfortunately they did no such thing so all Almeida was left with was unpurified organ and tissue culture goo to search through until she found and picked out the particles which looked like the “viruses” she wanted to see. That these particles not only resembled influenza but were rejected by peer review for being described as “bad images of influenza” should be enough to realize that all Almeida did was seek out “virus-like” particles fitting a preconceived mold of what a “virus” should look like. That these particles also magically happened to match those she had seen before in mice and chickens is further proof that Almeida relied on her own interpretation biases to label unpurified particles as the representation of these new “viruses.” Almeida did not discover any new “virus” in the unpurified goo given to her by Tyrrell and Hamre. All she did was image and resell the same particles previously identified as influenza back to the public as a scary new boogeyman now associated with the common cold.

3 Responses
locus
Scientific studies are extremely prone to shameless counterfeiting. Anyone who uses his sense of reality could ask endless questions about the deceptions of the so-called so-called scientific studies in the field of so-called molecular biology.
Petteri Sahinjoki
Electron beam is very hot. So – electron microscope images just show burned beef.
Read This;
https://news.mit.edu/2009/electron-microscope
HERMAN
all this does NOT mean that it is a virus, it could be just a fragment of some protein… and even if it is a virus, there is no evidence of its pathogenicity-harmfulness…. why don’t we accuse the firemen of being responsible for the fire just because, because we found them at the incinerator….pathogenicity must be proven… where are Koch’s postulates??